Amies Type, Stuart Type, and Cary-Blair Transport Media Products Introduction
Release time:
Mar 22,2024
Cida (Guangzhou) Biotechnology Co., Ltd. is located in the beautiful flower city-Guangzhou City. It is a high-tech enterprise integrating R & D, production and service of medical consumables. The company has passed ISO 13485 quality management system certification and EU CE certification. The company's products are exported to many countries and regions around the world, and has become a supplier in this field in South China.
CIDA (Guangzhou) Biotechnology Co., Ltd.
Amies Type, Stuart Type, and Cary-Blair Transport Media Products Introduction
Ⅰ.What is Transport Medium?
Transport medium is a medium used for the collection, transportation, and preservation of pathogenic bacteria or microbial specimens, including both liquid and solid types. It is generally used for the transportation and preservation of clinical specimen collections. It is provided to medical units for pretreatment of pathogenic tests to obtain pathogenic bacterial specimens.


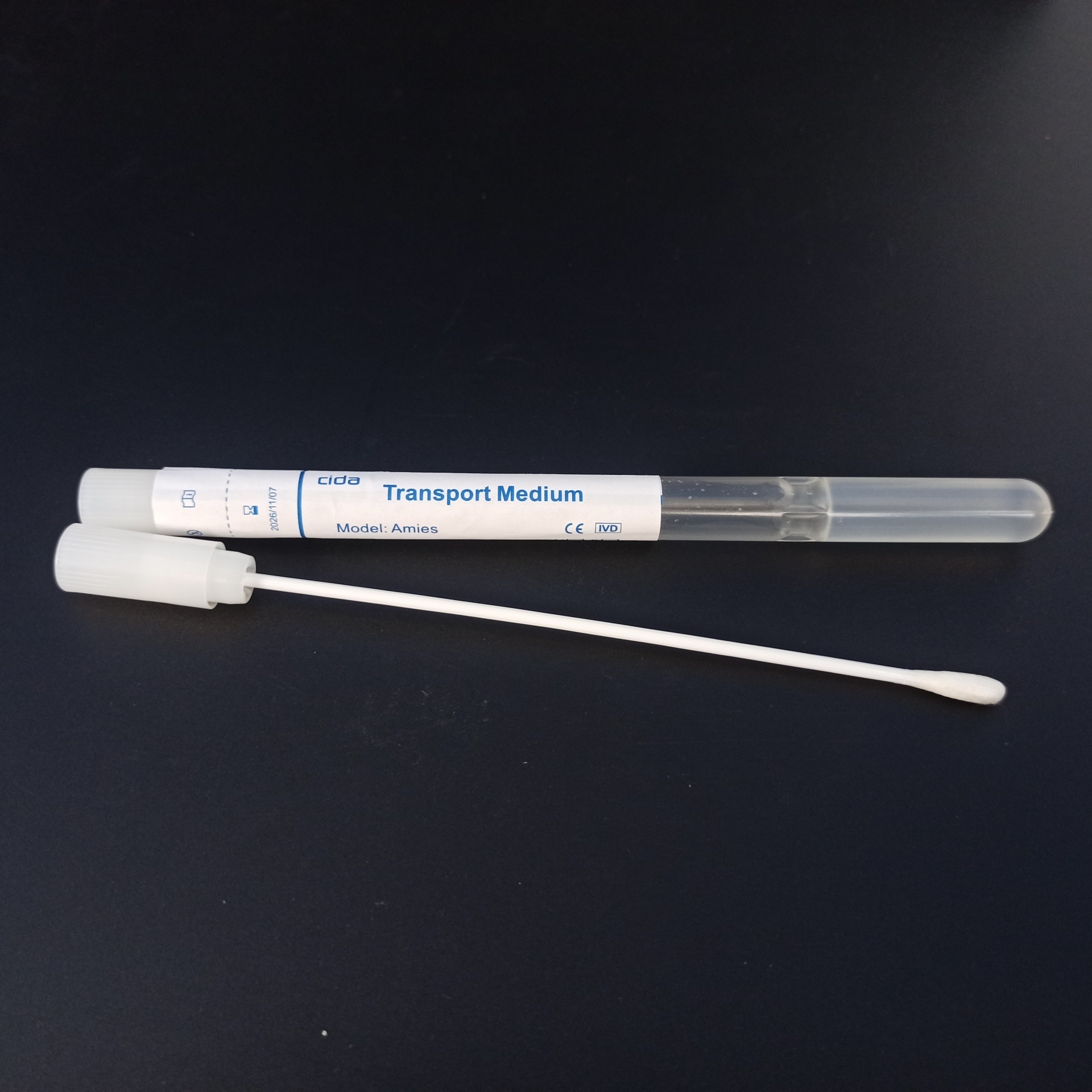
II. The Role of Transport Medium
Through different formulations of various transport mediums, the viability of the corresponding microorganisms in the specimens can be improved and enhanced for short-term storage during transportation. This maintains the activity of pathogenic bacteria without causing death, and does not contain enrichment components, allowing the specimen to be properly preserved with the microorganisms intact throughout the transportation process.



III. Product Features
1.Ready-to-use, easy and quick to operate, which improves the culture isolation rate of clinical pathogenic bacteria.
2.Available in both carbon-containing and carbon-free types to meet the needs of different bacteria.
3.Wide application range, suitable for oral, ophthalmology-otolaryngology, intestinal, urinary and genital tract, wound, and fecal bacterial tests.
4.Ensures the survival of a wide range of microorganisms for 24-48 hours at room temperature.
5.Specialized culture medium formulations for various specific pathogenic bacteria in different samples.
6.The overall design has a good effect on the transportation and storage of various clinical pathogenic bacteria.
7.Transport mediums are individually packed in aseptic packaging, convenient for clinical use.
8.Swabs for collecting samples have good adsorption capacity for various pathogens, effectively avoiding residual samples in the transport medium during transfer, and almost transferring all the samples on the swab head.


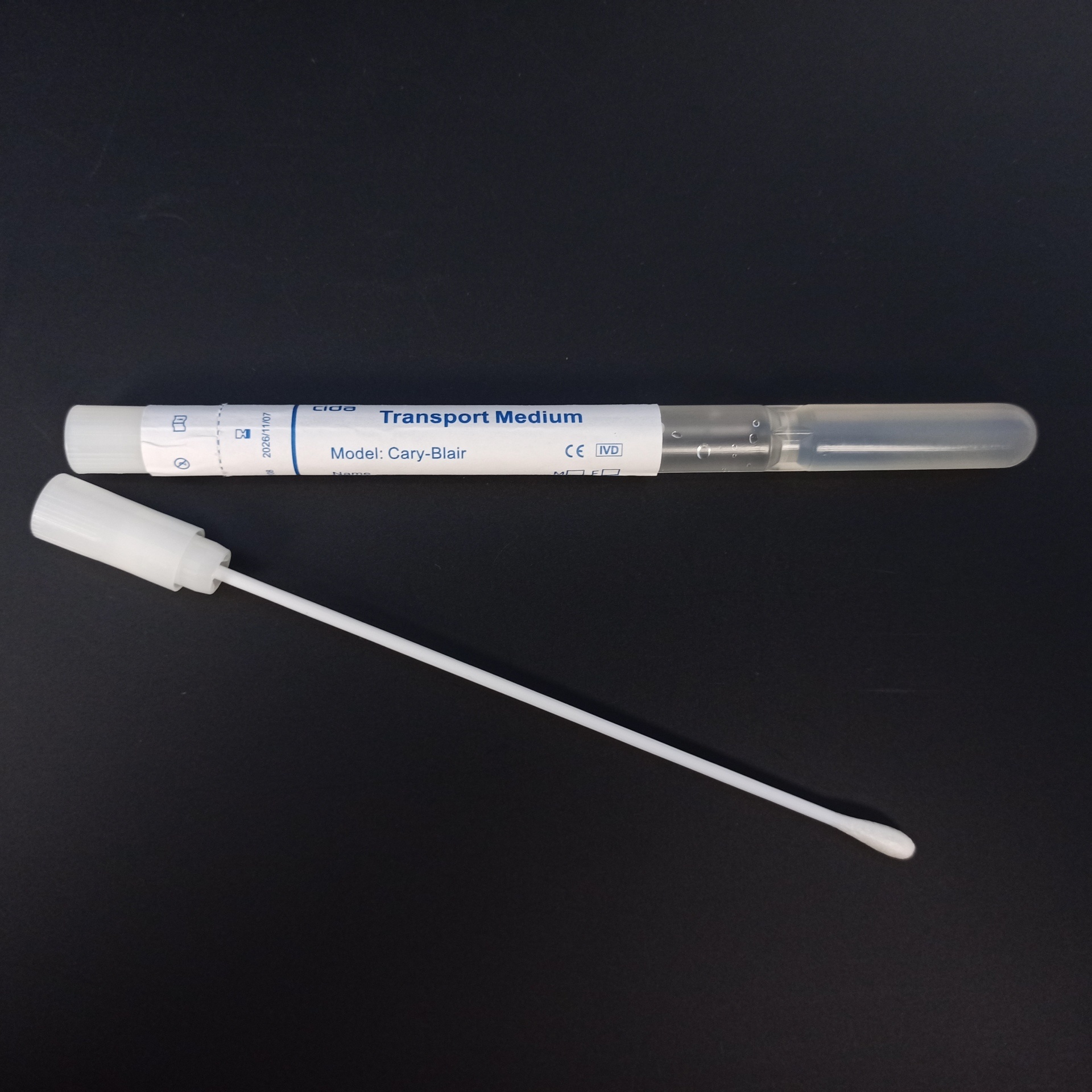
IV. Product Operation Demonstration and Precautions
Operation Method:
1.Remove the paper-plastic or aluminum-plastic packaging, take out the swab cotton swab, and collect the sample.
2.Remove the cap from the disposable transport medium tube and set it aside.
3.Insert the cotton swab containing the collected sample into the tube immediately and tightly press the tube cap.
4.Properly place and store the culture medium tube after sampling, and deliver it to the laboratory within 48 hours.
Precautions:
1.Strict aseptic techniques must be followed, and it is recommended to handle the collected samples within a biosafety cabinet or other protective devices.
2.Care should be taken when removing the sample swab from the transport tube to avoid splattering or generating aerosols.
3.The use of the product is strictly prohibited if the packaging is damaged. Used swab samples and transport tubes should be disposed of in accordance with the laboratory’s regulations for infectious waste.
Get a free quote!
More information
